The Break-down |
Molecular lasers and its break-down
|
Molecular lasers function differently than atomic lasers because they have vibrational and rotational energies as well as electronic energy. These molecular vibrations occur because the relative positions and orientations of the atomic nuclei are not absolutely fixed within the molecule. The molecular rotations occur because the individual molecules are free to spin and rotate in space since they are in a gaseous state. The energies associated with molecular vibrations and rotations are quantized just like the electronic energy. Transitions between vibrational energy levels emit photons with wavelengths in the infrared region, while transitions between rotational energy levels emit photons in the microwave region. The CO2 laser is based on the vibrational and rotational transitions of the CO2 molecule. This molecule consists of two oxygen atoms covalently bonded with a central carbon atom. Arranged in sp2 hybrid orbitals, each oxygen atom forms a sigma and a pi bond with the carbon atom and leaves two free electron pairs (Zumdahl, 639):
The dynamics of the carbon dioxide molecule are very similar to the dynamics of diatomic molecules such as CO or HCl.Molecular Vibration: In a diatomic molecule such as O2, N2 or CO, the individual atoms are bound by a molecular binding force that functions much like the spring constant k of a linear harmonic oscillator. When excited, the two nuclei will vibrate much like two masses connected by a spring. While real diatomic molecules are not perfect harmonic oscillators, their potential energy functions approximate those of a harmonic oscillator for a certain value of inter-nuclear separation. Although Carbon Dioxide is a triatomic molecule, it behaves much like a simple diatomic molecule because its structure is linear. Such a linear triatomic molecule has three normal modes of vibration, described as the asymmetric stretch mode, the bending mode and the symmetric stretch mode. Each one of these normal modes of vibration for the CO2 molecule is associated with a characteristic frequency of vibration (w) as well as a ladder of allowed energy levels. Therefore we label the three vibrational modes n1, n2, and n3, with their vibrational energy levels being quantized (n= 0, 1, 2, 3, . . .). To describe the vibrational modes we use the notation ( n1, n2, n3). We can approximate the vibrational energy levels as the quantum mechanical simple harmonic oscillator (hb= h/(2pi)):Ev=hbw0*(n+1/2), where v0 is the classical vibration frequencyw0= (k/u)^1/2Notice that there will be a ground state vibrational level (corresponding to n=0) with some positive energy E0= hbw/2 While in reality each normal mode is slightly anharmonic, the specific equation for the vibrational energy of CO2 can still be described by:E(n1, n2, n3) = hcw1 (n1+1/2) + hcw2 (n2+1/2) + hcw3 (n3+1/2) The following diagram depicts the first few vibrational energy levels (n1 n2 n3) of the CO2 molecule and also lists the normal modes' characteristic frequencies in units of wave numbers (cm-1). Notice that the base frequency for the symmetric stretch mode is w1=1288cm-1 and for the bending mode w2= 667cm-1. For the asymmetric stretch mode, the base frequency is much higher at w3= 2349cm-1.Molecular Rotation: Again because CO2 is a linear triatomic molecule and thus behaves very much like a diatomic molecule, it is considerably simple to understand the dynamics of its rotation. To start, picture the molecule as a classical dumbbell with masses on the ends connected by a rod. When the dumbbell then rotates about its center of mass (carbon atom) it has a kinetic energy that is a function of its moment of inertia and the length of the rod. This model must be adjusted somewhat to describe a diatomic molecule because the separation of the masses is not fixed. As the molecule rotates the oxygen atoms are pulled farther apart by the centrifugal force, therefore increasing the moment of inertia. It is even more complicated if the molecule is experiencing a molecular vibration. Fortunately, for our purposes the rigid dumbbell approximation will suffice for describing the rotational energy of a diatomic molecule. When the classical equation for rotational energy is put into a quantum mechanical form, with the quantum number J describing the rotational states, we then have an expression for the rotational energy of a diatomic molecule. Thanks to its linear structure, the rotational energy spectrum of CO2 has the same character as that for diatomic molecules and the rotational energy levels are thus approximated by: EJ = hcBeJ(J+1), J = 0, 1, 2, 3, ... where the rotational constant Be for the CO2 molecule is Be = .39 cm-1. The difference between energy levels is: |
scalar physical quantity
In physics, energy is a property of objects, transferable among them via fundamental interactions, which can beconverted into different forms but not created or destroyed. The joule is the SI unit of energy, based on the amount transferred to an object by the mechanical work of moving it 1 metre against a force of 1 newton.[1]
Work and heat are two categories of processes or mechanisms that can transfer a given amount of energy. Thesecond law of thermodynamics limits the amount of work that can be performed by energy that is obtained via a heating process—some energy is always lost as waste heat. The maximum amount that can go into work is called the available energy. Systems such as machines and living things often require available energy, not just any energy. Mechanical and other forms of energy can be transformed in the other direction into thermal energy without such limitations.
There are many forms of energy, but all these types must meet certain conditions such as being convertible to other kinds of energy, obeying conservation of energy, and causing a proportional change in mass in objects that possess it. Common energy forms include the kinetic energy of a moving object, the radiant energy carried by light and other electromagnetic radiation, the potential energy stored by virtue of the position of an object in a force field such as a gravitational, electric or magnetic field, and the thermal energy comprising the microscopic kinetic and potential energies of the disordered motions of the particles making up matter. Some specific forms of potential energy include elastic energy due to the stretching or deformation of solid objects and chemical energy such as is released when a fuel burns. Any object that has mass when stationary, such as a piece of ordinary matter, is said to have rest mass, or an equivalent amount of energy whose form is called rest energy, though this isn't immediately apparent in everyday phenomena described by classical physics.
According to mass–energy equivalence, all forms of energy (not just rest energy) exhibit mass. For example, adding 25 kilowatt-hours (90 megajoules) of energy to an object in the form of heat (or any other form) increases its mass by 1 microgram; if you had a sensitive enough mass balance or scale, this mass increase could be measured. Our Sun transforms nuclear potential energy to other forms of energy; its total mass does not decrease due to that in itself (since it still contains the same total energy even if in different forms), but its mass does decrease when the energy escapes out to its surroundings, largely as radiant energy.
Although any energy in any single form can be transformed into another form, the law of conservation of energy states that the total energy of a system can only change if energy is transferred into or out of the system. This means that it is impossible to create or destroy energy. The total energy of a system can be calculated by adding up all forms of energy in the system. Examples of energy transfer and transformation include generating or making use of electric energy, performingchemical reactions, or lifting an object. Lifting against gravity performs work on the object and stores gravitational potential energy; if it falls, gravity does work on the object which transforms the potential energy to the kinetic energy associated with its speed.
More broadly, living organisms require available energy to stay alive; humans get such energy from food along with the oxygen needed to metabolize it. Civilisation requires a supply of energy to function; energy resources such as fossil fuels are a vital topic in economics and politics. Earth's climate and ecosystem are driven by the radiant energy Earth receives from the sun (as well as the geothermal energy contained within the earth), and are sensitive to changes in the amount received. The word "energy" is also used outside of physics in many ways, which can lead to ambiguity and inconsistency. The vernacular terminology is not consistent withtechnical terminology. For example, while energy is always conserved (in the sense that the total energy does not change despite energy transformations), energy can be converted into a form, e.g., thermal energy, that cannot be utilized to perform work. When one talks about "conserving energy by driving less", one talks about conserving fossil fuels and preventing useful energy from being lost as heat. This usage of "conserve" differs from that of the law of conservation of energy
Work and heat are two categories of processes or mechanisms that can transfer a given amount of energy. Thesecond law of thermodynamics limits the amount of work that can be performed by energy that is obtained via a heating process—some energy is always lost as waste heat. The maximum amount that can go into work is called the available energy. Systems such as machines and living things often require available energy, not just any energy. Mechanical and other forms of energy can be transformed in the other direction into thermal energy without such limitations.
There are many forms of energy, but all these types must meet certain conditions such as being convertible to other kinds of energy, obeying conservation of energy, and causing a proportional change in mass in objects that possess it. Common energy forms include the kinetic energy of a moving object, the radiant energy carried by light and other electromagnetic radiation, the potential energy stored by virtue of the position of an object in a force field such as a gravitational, electric or magnetic field, and the thermal energy comprising the microscopic kinetic and potential energies of the disordered motions of the particles making up matter. Some specific forms of potential energy include elastic energy due to the stretching or deformation of solid objects and chemical energy such as is released when a fuel burns. Any object that has mass when stationary, such as a piece of ordinary matter, is said to have rest mass, or an equivalent amount of energy whose form is called rest energy, though this isn't immediately apparent in everyday phenomena described by classical physics.
According to mass–energy equivalence, all forms of energy (not just rest energy) exhibit mass. For example, adding 25 kilowatt-hours (90 megajoules) of energy to an object in the form of heat (or any other form) increases its mass by 1 microgram; if you had a sensitive enough mass balance or scale, this mass increase could be measured. Our Sun transforms nuclear potential energy to other forms of energy; its total mass does not decrease due to that in itself (since it still contains the same total energy even if in different forms), but its mass does decrease when the energy escapes out to its surroundings, largely as radiant energy.
Although any energy in any single form can be transformed into another form, the law of conservation of energy states that the total energy of a system can only change if energy is transferred into or out of the system. This means that it is impossible to create or destroy energy. The total energy of a system can be calculated by adding up all forms of energy in the system. Examples of energy transfer and transformation include generating or making use of electric energy, performingchemical reactions, or lifting an object. Lifting against gravity performs work on the object and stores gravitational potential energy; if it falls, gravity does work on the object which transforms the potential energy to the kinetic energy associated with its speed.
More broadly, living organisms require available energy to stay alive; humans get such energy from food along with the oxygen needed to metabolize it. Civilisation requires a supply of energy to function; energy resources such as fossil fuels are a vital topic in economics and politics. Earth's climate and ecosystem are driven by the radiant energy Earth receives from the sun (as well as the geothermal energy contained within the earth), and are sensitive to changes in the amount received. The word "energy" is also used outside of physics in many ways, which can lead to ambiguity and inconsistency. The vernacular terminology is not consistent withtechnical terminology. For example, while energy is always conserved (in the sense that the total energy does not change despite energy transformations), energy can be converted into a form, e.g., thermal energy, that cannot be utilized to perform work. When one talks about "conserving energy by driving less", one talks about conserving fossil fuels and preventing useful energy from being lost as heat. This usage of "conserve" differs from that of the law of conservation of energy
Gravitational potential energy
The gravitational force very near the surface of a massive body (e.g. a planet) varies very little with small changes in height, h, and locally is equal mg where m ismass, and g is the gravitational acceleration (AKA field strength). At the Earth's surface g = 9.81 m s-1. In these cases, the gravitational potential energy is given by
A more general expression for the potential energy due to Newtonian gravitation between two bodies of masses m1 and m2, is
,where r is the separation between the two bodies and G is the gravitational constant, 6.6742(10) × 10-11 m3 kg-1 s-2.[1] In this case, the zero potential reference point is the infinite separation of the two bodies. Care must be taken that these masses are point masses or uniform spherical solids/shells. It cannot be applied directly to any objects of any shape and any mass.
In terms of the gravitational potential (Φ, U or V), the potential energy is (by definition of gravitational potential),
.
A more general expression for the potential energy due to Newtonian gravitation between two bodies of masses m1 and m2, is
,where r is the separation between the two bodies and G is the gravitational constant, 6.6742(10) × 10-11 m3 kg-1 s-2.[1] In this case, the zero potential reference point is the infinite separation of the two bodies. Care must be taken that these masses are point masses or uniform spherical solids/shells. It cannot be applied directly to any objects of any shape and any mass.
In terms of the gravitational potential (Φ, U or V), the potential energy is (by definition of gravitational potential),
.
Thermal energy
General scope
Thermal energy (of some state of matter - gas, plasma, solid, etc.) is the energy associated with the microscopical random motion of particles constituting the media. For example, in case of monatomic gas it is just a kinetic energy of motion of atoms of gas as measured in the reference frame of the center of mass of gas. In case of molecules in the gas rotational and vibrational energy is involved. In the case of liquids and solids there is also potential energy (of interaction of atoms) involved, and so on.
A heat is defined as a transfer (flow) of thermal energy across certain boundary (for example, from a hot body to cold via the area of their contact). A practical definition for small transfers of heat is
where Cv is the heat capacity of the system. This definition will fail if the system undergoes a phase transition—e.g. if ice is melting to water—as in these cases the system can absorb heat without increasing its temperature. In more complex systems, it is preferable to use the concept of internal energy rather than that of thermal energy (see Chemical energy below).
Despite the theoretical problems, the above definition is useful in the experimental measurement of energy changes. In a wide variety of situations, it is possible to use the energy released by a system to raise the temperature of another object, e.g. a bath of water. It is also possible to measure the amount of electric energy required to raise the temperature of the object by the same amount. The calorie was originally defined as the amount of energy required to raise the temperature of one gram of water by 1 °C (approximately 4.1855 J, although the definition later changed), and the British thermal unitwas defined as the energy required to heat one pound of water by 1 °F (later fixed as 1055.06 J).
Kinetic theory
In kinetic theory which describes the ideal gas, the thermal energy per degree of freedom is given by:
where df is the number of degrees of freedom and kB is the Boltzmann constant. The total themeral energies would equal the total internal energy of the gas, since intermolecular potential energy is neglected in this theory. The term kBT occurs very frequently into statistical thermodynamics
Thermal energy (of some state of matter - gas, plasma, solid, etc.) is the energy associated with the microscopical random motion of particles constituting the media. For example, in case of monatomic gas it is just a kinetic energy of motion of atoms of gas as measured in the reference frame of the center of mass of gas. In case of molecules in the gas rotational and vibrational energy is involved. In the case of liquids and solids there is also potential energy (of interaction of atoms) involved, and so on.
A heat is defined as a transfer (flow) of thermal energy across certain boundary (for example, from a hot body to cold via the area of their contact). A practical definition for small transfers of heat is
where Cv is the heat capacity of the system. This definition will fail if the system undergoes a phase transition—e.g. if ice is melting to water—as in these cases the system can absorb heat without increasing its temperature. In more complex systems, it is preferable to use the concept of internal energy rather than that of thermal energy (see Chemical energy below).
Despite the theoretical problems, the above definition is useful in the experimental measurement of energy changes. In a wide variety of situations, it is possible to use the energy released by a system to raise the temperature of another object, e.g. a bath of water. It is also possible to measure the amount of electric energy required to raise the temperature of the object by the same amount. The calorie was originally defined as the amount of energy required to raise the temperature of one gram of water by 1 °C (approximately 4.1855 J, although the definition later changed), and the British thermal unitwas defined as the energy required to heat one pound of water by 1 °F (later fixed as 1055.06 J).
Kinetic theory
In kinetic theory which describes the ideal gas, the thermal energy per degree of freedom is given by:
where df is the number of degrees of freedom and kB is the Boltzmann constant. The total themeral energies would equal the total internal energy of the gas, since intermolecular potential energy is neglected in this theory. The term kBT occurs very frequently into statistical thermodynamics
Chemical energy
Chemical energy is the energy due to excretion of atoms in molecules and various other kinds of aggregates of matter. It may be defined as a work done by electric forces during re-arrangement of mutual positions of electric charges, electrons and protons, in the process of aggregation. So, basically it is electrostatic potential energy of electric charges. If the chemical energy of a system decreases during a chemical reaction, the difference is transferred to the surroundings in some form (often heat or light); on the other hand if the chemical energy of a system increases as a result of a chemical reaction - the difference then is supplied by the surroundings (usually again in form of heat or light). For example,
when two hydrogen atoms react to form a dihydrogen molecule, the chemical energy decreases by 724 zJ (the bond energyof the H–H bond);when the electron is completely removed from a hydrogen atom, forming a hydrogen ion (in the gas phase), the chemical energy increases by 2.18 aJ (the ionization energy of hydrogen).It is common to quote the changes in chemical energy for one mole of the substance in question: typical values for the change in molar chemical energy during a chemical reaction range from tens to hundreds of kilojoules per mole.
The chemical energy as defined above is also referred to by chemists as the internal energy, U: technically, this is measured by keeping the volume of the system constant. Most practical chemistry is performed at constant pressure and, if the volume changes during the reaction (e.g. a gas is given off), a correction must be applied to take account of the work done by or on the atmosphere to obtain the enthalpy, H, this correction is the work done by an expanding gas,
,so the enthalpy now reads;
.A second correction, for the change in entropy, S, must also be performed to determine whether a chemical reaction will take place or not, giving the Gibbs free energy, G. The correction is the energy required to create order from disorder,[2]
,so we have;
.These corrections are sometimes negligible, but often not (especially in reactions involving gases).
Since the industrial revolution, the burning of coal, oil, natural gas or products derived from them has been a socially significant transformation of chemical energy into other forms of energy. the energy "consumption" (one should really speak of "energy transformation") of a society or country is often quoted in reference to the average energy released by the combustion of these fossil fuels:
1 tonne of coal equivalent (TCE) = 29.3076 GJ = 8,141 kilowatt hour1 tonne of oil equivalent (TOE) = 41.868 GJ = 11,630 kilowatt hourOn the same basis, a tank-full of gasoline (45 litres, 12 gallons) is equivalent to about 1.6 GJ of chemical energy. Another chemically based unit of measurement for energy is the "tonne of TNT", taken as 4.184 GJ. Hence, burning a tonne of oil releases about ten times as much energy as the explosion of one tonne of TNT: fortunately, the energy is usually released in a slower, more controlled manner.
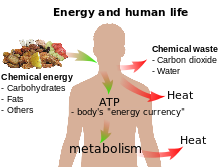
Simple examples of storage of chemical energy are batteries and food. When food is digested and metabolized (often with oxygen), chemical energy is released, which can in turn be transformed into heat, or by muscles into kinetic energy.
According to the Bohr theory of the atom, the chemical energy is characterized by the Rydberg constant.
when two hydrogen atoms react to form a dihydrogen molecule, the chemical energy decreases by 724 zJ (the bond energyof the H–H bond);when the electron is completely removed from a hydrogen atom, forming a hydrogen ion (in the gas phase), the chemical energy increases by 2.18 aJ (the ionization energy of hydrogen).It is common to quote the changes in chemical energy for one mole of the substance in question: typical values for the change in molar chemical energy during a chemical reaction range from tens to hundreds of kilojoules per mole.
The chemical energy as defined above is also referred to by chemists as the internal energy, U: technically, this is measured by keeping the volume of the system constant. Most practical chemistry is performed at constant pressure and, if the volume changes during the reaction (e.g. a gas is given off), a correction must be applied to take account of the work done by or on the atmosphere to obtain the enthalpy, H, this correction is the work done by an expanding gas,
,so the enthalpy now reads;
.A second correction, for the change in entropy, S, must also be performed to determine whether a chemical reaction will take place or not, giving the Gibbs free energy, G. The correction is the energy required to create order from disorder,[2]
,so we have;
.These corrections are sometimes negligible, but often not (especially in reactions involving gases).
Since the industrial revolution, the burning of coal, oil, natural gas or products derived from them has been a socially significant transformation of chemical energy into other forms of energy. the energy "consumption" (one should really speak of "energy transformation") of a society or country is often quoted in reference to the average energy released by the combustion of these fossil fuels:
1 tonne of coal equivalent (TCE) = 29.3076 GJ = 8,141 kilowatt hour1 tonne of oil equivalent (TOE) = 41.868 GJ = 11,630 kilowatt hourOn the same basis, a tank-full of gasoline (45 litres, 12 gallons) is equivalent to about 1.6 GJ of chemical energy. Another chemically based unit of measurement for energy is the "tonne of TNT", taken as 4.184 GJ. Hence, burning a tonne of oil releases about ten times as much energy as the explosion of one tonne of TNT: fortunately, the energy is usually released in a slower, more controlled manner.
Simple examples of storage of chemical energy are batteries and food. When food is digested and metabolized (often with oxygen), chemical energy is released, which can in turn be transformed into heat, or by muscles into kinetic energy.
According to the Bohr theory of the atom, the chemical energy is characterized by the Rydberg constant.
Electric energy
Electrostatic energy[edit]General scope
The electric potential energy of given configuration of charges is defined as the work which must be done against the Coulomb force to rearrange charges from infinite separation to this configuration (or the work done by the Coulomb force separating the charges from this configuration to infinity). For two point-like charges Q1 and Q2 at a distance r this work, and hence electric potential energy is equal to:
where ε0 is the electric constant of a vacuum, 107/4πc02 or 8.854188… × 10−12 F m−1.[1] In terms of electrostatic potential (ϕ for absolute, V for difference in potential), again by definition, electrostatic potential energy is given by:
.If the charge is accumulated in a capacitor (of capacitance C), the reference configuration is usually selected not to be infinite separation of charges, but vice versa - charges at an extremely close proximity to each other (so there is zero net charge on each plate of a capacitor). The justification for this choice is purely practical - it is easier to measure both voltage difference and magnitude of charges on a capacitor plates not versus infinite separation of charges but rather versus discharged capacitor where charges return to close proximity to each other (electrons and ions recombine making the plates neutral). In this case the work and thus the electric potential energy becomes
,(different forms obtained using the definition of capacitance).
Electric energy[edit]Main article: Electric energy
Electric circuits
If an electric current passes through a resistor, electric energy is converted to heat; if the current passes through an electric appliance, some of the electric energy will be converted into other forms of energy (although some will always be lost as heat). The amount of electric energy due to an electric current can be expressed in a number of different ways:
where V is the electric potential difference (in volts), Q is the charge (in coulombs), I is the current (in amperes), t is the time for which the current flows (in seconds),P is the power (in watts) and R is the electric resistance (in ohms). The last of these expressions is important in the practical measurement of energy, as potential difference, resistance and time can all be measured with considerable accuracy.
Magnetic energy[edit]Main article: Magnetic energy
General scope
There is no fundamental difference between magnetic energy and electric energy: the two phenomena are related by Maxwell's equations. The potential energy of a magnet of magnetic moment m in a magnetic field B is defined as the work of magnetic force (actually of magnetic torque) on re-alignment of the vector of the magnetic dipole moment, and is equal to:
.Electric circuits
The energy stored in an inductor (of inductance L) carrying current I is
.This second expression forms the basis for superconducting magnetic energy storage.
Electromagnetic energy[edit]Examples of the interconversion of energyElectromagnetic radiation is convertedintobyMechanical energySolar sailThermal energySolar collectorElectric energySolar cellElectromagnetic radiationNon-linear opticsChemical energyPhotosynthesisNuclear energyMössbauer spectroscopyCalculating work needed to create an electric or magnetic field in unit volume (say, in a capacitor or an inductor) results in the electric and magnetic fields energy densities:
,in SI units.
Electromagnetic radiation, such as microwaves, visible light or gamma rays, represents a flow of electromagnetic energy. Applying the above expressions to magnetic and electric components of electromagnetic field both the volumetric density and the flow of energy in EM field can be calculated. The resulting Poynting vector, which is expressed as
in SI units, gives the density of the flow of energy and its direction.
The energy of electromagnetic radiation is quantized (has discrete energy levels). The energy of a photon is:
,so the spacing between energy levels is:
,where h is the Planck constant, 6.6260693(11)×10−34 Js,[1] and ν is the frequency of the radiation. This quantity of electromagnetic energy is usually called a photon. The photons which make up visible light have energies of 270–520 yJ, equivalent to 160–310 kJ/mol, the strength of weaker chemical bonds.
The electric potential energy of given configuration of charges is defined as the work which must be done against the Coulomb force to rearrange charges from infinite separation to this configuration (or the work done by the Coulomb force separating the charges from this configuration to infinity). For two point-like charges Q1 and Q2 at a distance r this work, and hence electric potential energy is equal to:
where ε0 is the electric constant of a vacuum, 107/4πc02 or 8.854188… × 10−12 F m−1.[1] In terms of electrostatic potential (ϕ for absolute, V for difference in potential), again by definition, electrostatic potential energy is given by:
.If the charge is accumulated in a capacitor (of capacitance C), the reference configuration is usually selected not to be infinite separation of charges, but vice versa - charges at an extremely close proximity to each other (so there is zero net charge on each plate of a capacitor). The justification for this choice is purely practical - it is easier to measure both voltage difference and magnitude of charges on a capacitor plates not versus infinite separation of charges but rather versus discharged capacitor where charges return to close proximity to each other (electrons and ions recombine making the plates neutral). In this case the work and thus the electric potential energy becomes
,(different forms obtained using the definition of capacitance).
Electric energy[edit]Main article: Electric energy
Electric circuits
If an electric current passes through a resistor, electric energy is converted to heat; if the current passes through an electric appliance, some of the electric energy will be converted into other forms of energy (although some will always be lost as heat). The amount of electric energy due to an electric current can be expressed in a number of different ways:
where V is the electric potential difference (in volts), Q is the charge (in coulombs), I is the current (in amperes), t is the time for which the current flows (in seconds),P is the power (in watts) and R is the electric resistance (in ohms). The last of these expressions is important in the practical measurement of energy, as potential difference, resistance and time can all be measured with considerable accuracy.
Magnetic energy[edit]Main article: Magnetic energy
General scope
There is no fundamental difference between magnetic energy and electric energy: the two phenomena are related by Maxwell's equations. The potential energy of a magnet of magnetic moment m in a magnetic field B is defined as the work of magnetic force (actually of magnetic torque) on re-alignment of the vector of the magnetic dipole moment, and is equal to:
.Electric circuits
The energy stored in an inductor (of inductance L) carrying current I is
.This second expression forms the basis for superconducting magnetic energy storage.
Electromagnetic energy[edit]Examples of the interconversion of energyElectromagnetic radiation is convertedintobyMechanical energySolar sailThermal energySolar collectorElectric energySolar cellElectromagnetic radiationNon-linear opticsChemical energyPhotosynthesisNuclear energyMössbauer spectroscopyCalculating work needed to create an electric or magnetic field in unit volume (say, in a capacitor or an inductor) results in the electric and magnetic fields energy densities:
,in SI units.
Electromagnetic radiation, such as microwaves, visible light or gamma rays, represents a flow of electromagnetic energy. Applying the above expressions to magnetic and electric components of electromagnetic field both the volumetric density and the flow of energy in EM field can be calculated. The resulting Poynting vector, which is expressed as
in SI units, gives the density of the flow of energy and its direction.
The energy of electromagnetic radiation is quantized (has discrete energy levels). The energy of a photon is:
,so the spacing between energy levels is:
,where h is the Planck constant, 6.6260693(11)×10−34 Js,[1] and ν is the frequency of the radiation. This quantity of electromagnetic energy is usually called a photon. The photons which make up visible light have energies of 270–520 yJ, equivalent to 160–310 kJ/mol, the strength of weaker chemical bonds.